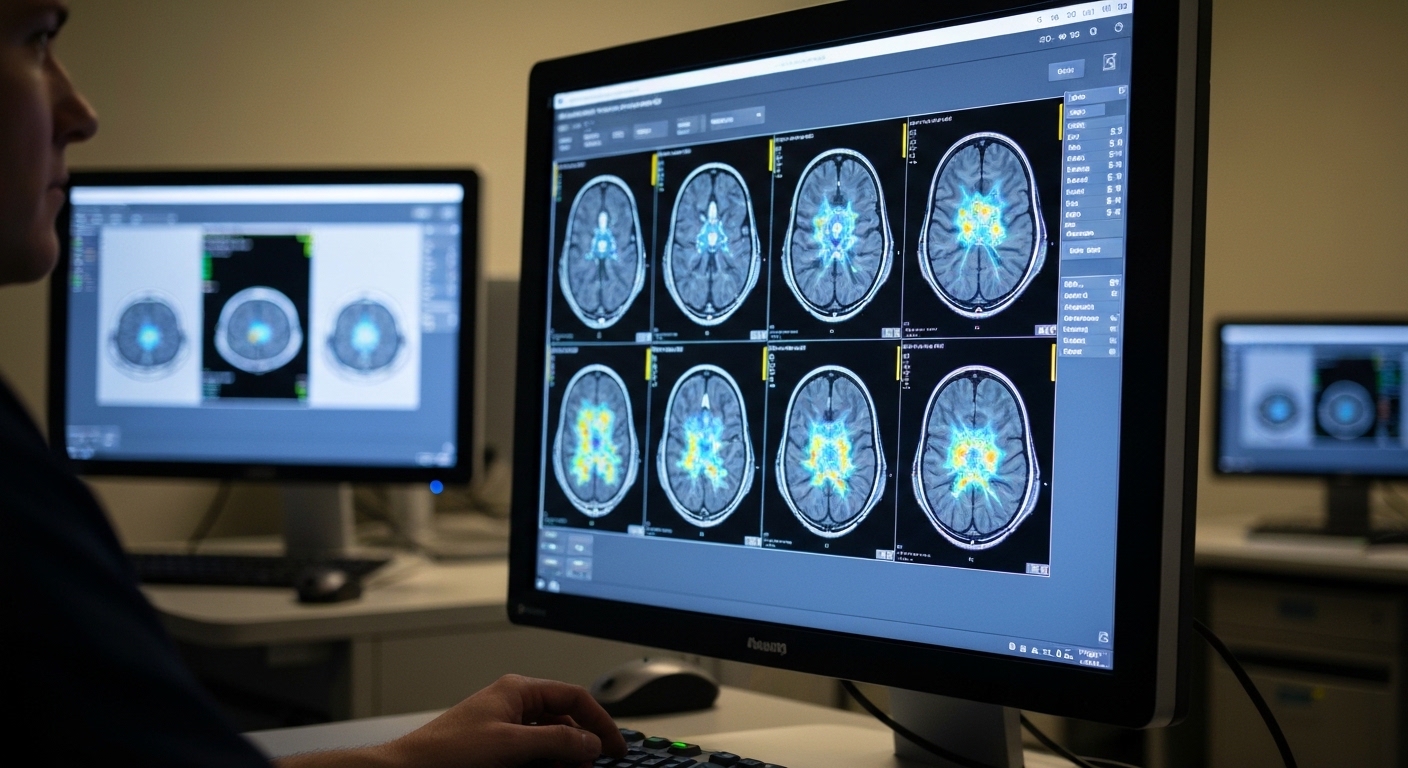
Sustained Attention Tasks During MRI Improve Prediction of Autistic Traits
Functional imaging during cognitive challenges identifies neural markers of social responsiveness more accurately than resting-state scans.
The Clinical Lighthouse
Already a member?
or
The Clinical Lighthouse
Subscribe to read the full analysis
Full access to every article, clinical summary, and the daily evidence brief.
- Unlimited access to all articles
- Daily evidence digest
- Daily podcast with news summaries
- Lighthouse Guideline Briefings
- Ask Lumi: your guide to search, summarise, and explain the archive
- Cancel anytime
