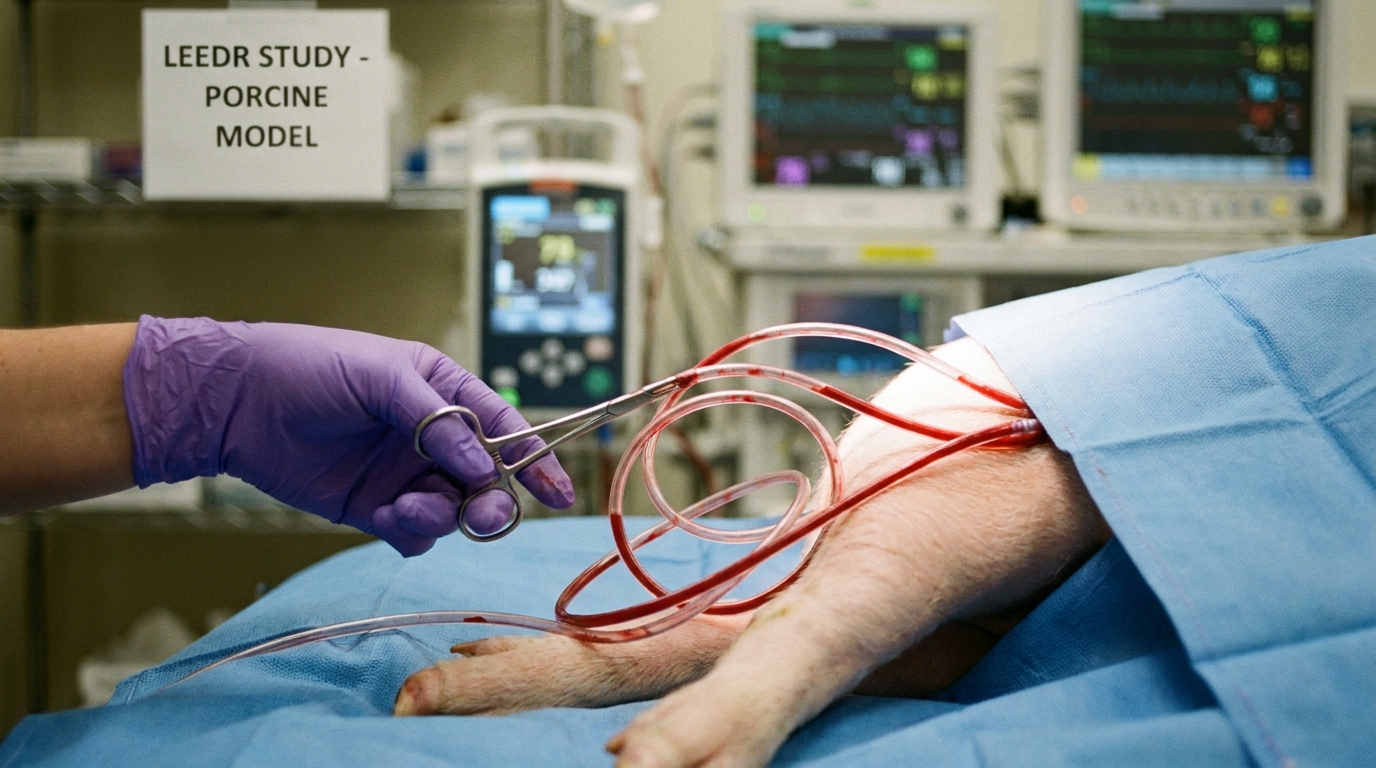
- Researchers investigated whether extracorporeal limb perfusion exacerbates hemorrhage in patients with uncontrolled vascular injuries following traumatic acute limb ischemia.
- This study utilized twelve swine with either isolated femoral arterial or combined arterial and venous injuries to evaluate extracorporeal distal revascularization.
- Post-perfusion blood loss did not differ significantly between groups, measuring zero milliliters versus 175 milliliters (p=0.34) at high pump speeds.
- The researchers concluded that extracorporeal perfusion did not meaningfully exacerbate hemorrhage, as histologic analysis confirmed that early thrombus remained intact.
- These findings suggest that vascular injury may be a relative contraindication to distal revascularization, warranting further clinical feasibility evaluations.
Bridging the Ischemic Gap in Traumatic Limb Salvage
Acute limb ischemia following traumatic injury represents a critical race against time, as prolonged warm ischemia rapidly leads to irreversible tissue necrosis and systemic metabolic complications. While standard revascularization remains the definitive treatment, delays in surgical intervention are frequent in complex trauma cases, necessitating strategies to maintain distal perfusion [1]. Meta-analytic data involving 2,040 patients show that prophylactic distal perfusion catheters (small-bore tubes placed to maintain blood flow distal to a large arterial cannula) significantly decrease the risk of acute limb ischemia (relative risk: 0.41; 95% confidence interval: 0.24 to 0.71) [2]. However, deploying these extracorporeal circuits in the setting of active hemorrhage remains a significant concern for clinicians, given that bleeding complications occur in approximately 49% of patients receiving venoarterial support [3, 4]. To address this clinical dilemma, researchers utilized a porcine model to evaluate whether Lower Extremity Extracorporeal Distal Revascularization (LEEDR), a technique that shunts arterial blood to an ischemic limb via an external pump, can be safely deployed when vascular integrity is compromised [5].
Simulating Vascular Trauma and Extracorporeal Support
To evaluate the hemorrhage risk associated with Lower Extremity Extracorporeal Distal Revascularization (LEEDR), investigators developed a standardized porcine model of groin hemorrhage. The study cohort included 12 Yorkshire swine divided into two distinct trauma profiles: six animals sustained a femoral arterial injury, and six sustained a combined femoral arterial and venous injury. This dual-group design allowed researchers to assess whether the addition of venous trauma alters the stability of hemostasis during extracorporeal perfusion. To simulate a severe clinical trauma event, the team created a 5-millimeter incision in the targeted vasculature, allowing for a controlled free bleeding volume of 300 mL. Following this initial hemorrhage, researchers achieved hemostasis using combat gauze, a standard kaolin-impregnated hemostatic dressing frequently used in trauma bays. Once bleeding was controlled, the team established percutaneous vascular access and initiated the LEEDR circuit. The system utilized contralateral femoral arterial inflow and distal saphenous artery outflow to bypass the injury site and deliver oxygenated blood to the distal limb. This setup directly tested whether the mechanical flow generated by an external pump would dislodge the fragile early thrombus formed at the vascular defect, a primary fear for surgeons considering extracorporeal support prior to definitive vascular repair.
Hemodynamic Stress and Hemorrhagic Stability
To assess the hemodynamic limits of the extracorporeal circuit and its impact on clot stability, the researchers implemented a rigorous stress test protocol. After initiating the perfusion circuit, the pump speed was increased in 1,000 revolutions per minute (RPM) increments every 10 minutes. This escalation continued until the system reached a programmed maximum pump speed of 8,000 RPM or until mechanical system limits were encountered. By artificially driving up the flow rate, the investigators could observe whether increased shear stress would dislodge the hemostatic plugs in the injured femoral vessels. The procedure proved highly feasible, as all 12 animals successfully underwent the full extracorporeal perfusion protocol. Mechanical performance was comparable across both injury models, with the femoral arterial injury group reaching a maximal speed of 6,708 ± 990 RPM and the combined arterial and venous injury group achieving 6,442 ± 1,107 RPM. Statistical analysis confirmed no significant difference in the maximal achieved RPM between the two groups (p=0.67), indicating that concomitant venous trauma did not mechanically limit the delivery of distal perfusion. Most importantly, the data revealed remarkable clot stability despite the absence of surgical vascular repair. In the isolated arterial injury group, no additional hemorrhage occurred during the perfusion protocol, resulting in an overall post-perfusion blood loss of 0 ± 0 mL. Within the combined injury group, one animal experienced additional bleeding at high RPM, which contributed to an overall post-perfusion blood loss of 175 ± 428.7 mL. Despite this single bleeding event, there was no statistically significant difference in post-perfusion blood loss between the two injury groups (p=0.34). These results demonstrate that an early, gauze-supported thrombus can largely withstand the hemodynamic pressures of distal revascularization.
Histologic Evidence of Clot Integrity
At the conclusion of the study, researchers performed a microscopic examination of the injured femoral vessels to evaluate the structural stability of the hemostatic plugs. This histologic analysis demonstrated intact early thrombus (the initial fibrin and platelet matrix formed at the injury site) in both arterial and venous injuries. Despite the significant mechanical stress introduced by the extracorporeal pump, histology showed no evidence of thrombus washout (the dislodging or erosion of a clot by the force of blood flow). These microscopic findings provide a clear structural explanation for the minimal secondary hemorrhage observed during the revascularization protocol. The investigators hypothesize that specific physiological factors protected the fragile clots from the high flow rates of the circuit. Specifically, distal arterial resistance (the natural opposition to blood flow within the smaller capillary beds of the limb) and favorable shear conditions (the frictional force exerted by blood flow against the vessel wall) may preserve thrombus integrity by buffering the full force of the pump before it reaches the vascular defect. From a clinical perspective, these findings suggest that uncontrolled vascular injury may represent a relative, rather than absolute, contraindication to extracorporeal distal revascularization. For the practicing trauma surgeon, this raises the possibility that limb salvage perfusion could be safely initiated to prevent irreversible ischemia even before definitive surgical repair is completed, provided that initial gauze-based hemostasis has been achieved.
References
1. Jongkind V, Earnshaw JJ, Gonçalves FB, et al. Editor's Choice – Update of the European Society for Vascular Surgery (ESVS) 2020 Clinical Practice Guidelines on the Management of Acute Limb Ischaemia in Light of the COVID-19 Pandemic, Based on a Scoping Review of the Literature. European Journal of Vascular and Endovascular Surgery. 2021. doi:10.1016/j.ejvs.2021.08.028
2. Gouchoe DA, Chaurasia S, Henn M, et al. Does Size Matter? The Effect of Size of Distal Perfusion Catheter on Acute Limb Ischemia: A Meta-Analysis. ASAIO journal (1992). 2024. doi:10.1097/MAT.0000000000002178
3. Rajsic S, Treml B, Jadzic D, et al. Extracorporeal membrane oxygenation for cardiogenic shock: a meta-analysis of mortality and complications. Annals of Intensive Care. 2022. doi:10.1186/s13613-022-01067-9
4. Coccolini F, Shander A, Ceresoli M, et al. Strategies to prevent blood loss and reduce transfusion in emergency general surgery, WSES-AAST consensus paper. World Journal of Emergency Surgery. 2024. doi:10.1186/s13017-024-00554-7
5. Dvir M, Nye K, Qadri S, et al. Characterizing bleeding risk of extracorporeal limb salvage with concomitant vascular injury.. The journal of trauma and acute care surgery. 2026. doi:10.1097/TA.0000000000005016