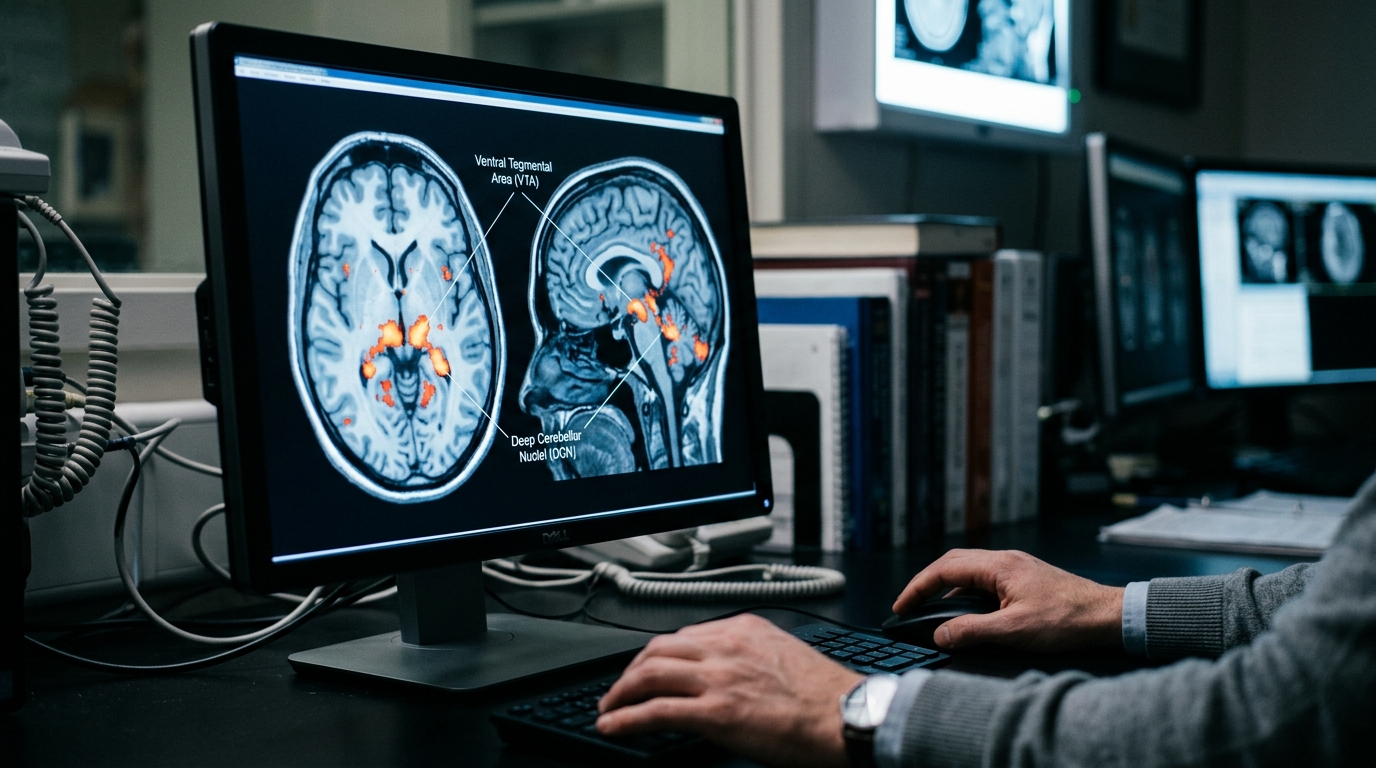
- The study investigated how nitrous oxide (N2O) influences ventral tegmental area (VTA) function to mediate reward behavior.
- Researchers observed conditioned place preference (CPP) with 60% N2O exposure, but not 30%, in an animal model.
- 60% N2O exposure increased excitability and glutamatergic transmission of VTA dopamine neurons and DCN activity.
- The authors concluded that N2O directly enhances AMPA receptor-mediated glutamatergic transmission in the DCN-VTA circuit.
- Attenuating N2O-induced potentiation of the DCN-VTA circuit may offer a therapeutic strategy for N2O-related reward behavior.
Understanding Nitrous Oxide's Addictive Potential
The recreational abuse of nitrous oxide has increased substantially worldwide, particularly among adolescents, creating significant public health concerns over its addictive potential [1, 2]. While long used in clinical practice as an anesthetic and analgesic, its capacity for misuse is now well-recognized, aligning it with other non-opioid anesthetics [3, 4]. Substance use disorders are chronic, relapsing conditions driven by persistent drug-seeking behaviors that impose a heavy burden on individuals and society [5]. Although current addiction therapies often aim to modulate maladaptive memories or target broad neurobiological systems, the specific mechanisms for many substances, including nitrous oxide, remain poorly defined [6, 7]. A recent study provides new clarity on the neurobiological underpinnings of nitrous oxide's rewarding effects, identifying a specific neural circuit that could inform more targeted interventions.
Identifying the Reward Pathway: VTA and N2O
To determine how nitrous oxide fosters addictive behavior, researchers focused on the ventral tegmental area (VTA), a midbrain structure central to the brain's reward circuitry and a key mediator of the effects of many drugs of abuse. The investigation began by establishing the drug's rewarding properties using a conditioned place preference model, a standard behavioral test where an animal's preference for an environment previously associated with a drug is measured. The findings revealed a clear dose-dependent effect: exposure to 60% nitrous oxide effectively induced reward-seeking behavior, whereas a 30% concentration did not. This behavioral outcome was linked to distinct physiological changes within the VTA. The study demonstrated that the 60% concentration simultaneously increased the excitability of VTA dopamine neurons and enhanced glutamatergic transmission to these same cells, indicating a direct impact on the activity and communication within this critical reward hub.
Unpacking the Molecular Mechanism: Beyond Homocysteine
Seeking the origin of the signals driving the ventral tegmental area (VTA), the researchers mapped upstream brain regions that provide input to it. Their analysis revealed that nitrous oxide specifically elevated homocysteine levels in the deep cerebellar nuclei (DCN), a region traditionally associated with motor control but increasingly implicated in cognitive and reward functions. This finding initially suggested a potential role for homocysteine in mediating the drug's effects. However, when the investigators pharmacologically blocked homocysteine production, they found it did not significantly reduce the drug-induced reward behavior or the heightened glutamatergic transmission to VTA neurons. This result indicated that while homocysteine levels were affected, they were not the primary driver of the addictive behavior.
Instead, the study uncovered a more direct molecular action. Despite nitrous oxide's known function as an N-methyl-D-aspartate (NMDA) receptor antagonist, the researchers discovered that it directly enhanced glutamatergic transmission mediated by AMPA receptors (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors). This potentiation of AMPA receptor signaling, a different class of glutamate receptor, functionally increased the activity of neurons in both the deep cerebellar nuclei and the VTA. This suggests a specific mechanism where nitrous oxide boosts the DCN-VTA circuit's activity by acting on AMPA receptors, thereby producing its rewarding effects.
Targeting the DCN-VTA Circuit for Intervention
Having identified the deep cerebellar nuclei (DCN) to ventral tegmental area (VTA) pathway as a key player, the study next sought to confirm its causal role in nitrous oxide-induced reward. Using advanced neuroscientific tools, the researchers demonstrated that weakening the connections within this specific circuit could reverse the drug's effects. Inducing long-term depression in the DCN-VTA pathway via optogenetics, a technique that uses light to precisely control neuronal activity, successfully attenuated the reward-seeking behavior. This finding confirms that the integrity and potentiation of this specific circuit are necessary for nitrous oxide's rewarding properties.
To further validate the VTA's central involvement, a separate experiment used chemogenetics, a method employing a designer drug to selectively silence specific cells. The results showed that chemogenetic inhibition of VTA dopamine neurons also diminished the nitrous oxide-induced reward behavior. Taken together, these experiments provide strong evidence that the DCN-VTA circuit is a critical mediator of nitrous oxide's addictive potential. For clinicians, these findings are highly relevant, as they move beyond a general understanding of reward to pinpoint a specific, modifiable pathway. The authors conclude that strategies aimed at attenuating the nitrous oxide-induced potentiation of the DCN-VTA circuit may represent a specific therapeutic approach for treating nitrous oxide-related substance use disorders.
References
1. Li W, Zhang Z. 263. NITROUS OXIDE EXERTS REWARDING EFFECT VIA REGULATING D1 RECEPTOR AND BDNF PATHWAY IN VENTRAL TEGMENTAL AREA-NUCLEUS ACCUMBENS DOPAMINE CIRCUIT. International Journal of Neuropsychopharmacology. 2025. doi:10.1093/ijnp/pyaf052.132
2. Back S, Kroon E, Colyer‐Patel K, Cousijn J. Does nitrous oxide addiction exist? An evaluation of the evidence for the presence and prevalence of substance use disorder symptoms in recreational nitrous oxide users. Addiction. 2023. doi:10.1111/add.16380
3. Sun Y, Sun X, Wu H, Xiao Z, W L. A review of recent advances in anesthetic drugs for patients undergoing cardiac surgery. Frontiers in Pharmacology. 2025. doi:10.3389/fphar.2025.1533162
4. Deng L, Wu L, Gao R, Xu X, Chen C, Liu J. Non-Opioid Anesthetics Addiction: A Review of Current Situation and Mechanism. Brain Sciences. 2023. doi:10.3390/brainsci13091259
5. Feltenstein MW, See R. The neurocircuitry of addiction: an overview. British Journal of Pharmacology. 2008. doi:10.1038/bjp.2008.51
6. Walsh K, Das R, Saladin ME, Kamboj SK. Modulation of naturalistic maladaptive memories using behavioural and pharmacological reconsolidation-interfering strategies: a systematic review and meta-analysis of clinical and ‘sub-clinical’ studies. Psychopharmacology. 2018. doi:10.1007/s00213-018-4983-8
7. McPartland JM, Guy GW, Marzo VD. Care and Feeding of the Endocannabinoid System: A Systematic Review of Potential Clinical Interventions that Upregulate the Endocannabinoid System. PLoS ONE. 2014. doi:10.1371/journal.pone.0089566