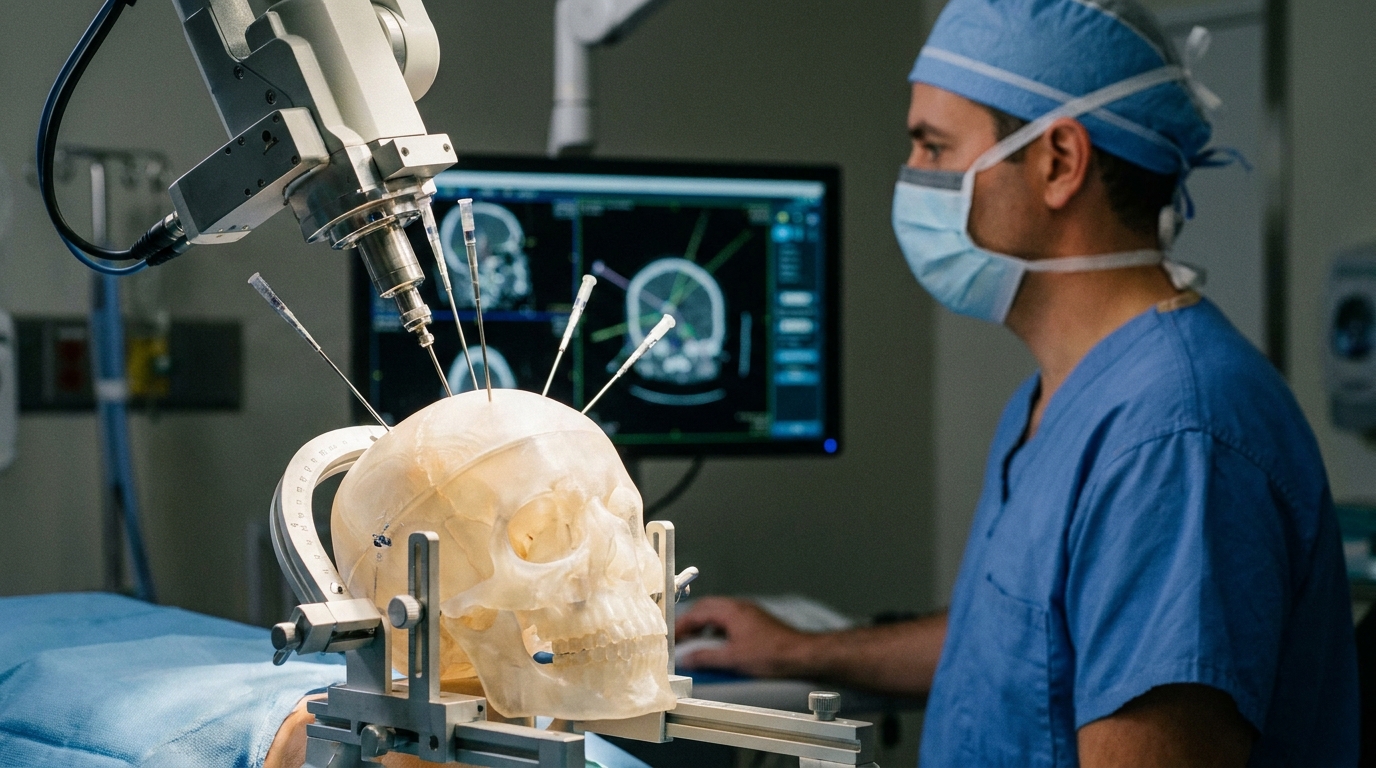
For Doctors in a Hurry
- Researchers investigated if cranial robotics with CT guidance could efficiently place multiple infusion cannulas for convection-enhanced delivery in brain tissue.
- The study utilized five 3D-printed phantom heads to test thirty bilateral targets using frontal, parietal, and occipital entry points.
- Placement achieved a mean radial error of 0.4 millimeters on CT and 0.6 millimeters on MRI within thirty minutes.
- The authors concluded that simultaneous robotic insertion of multiple cannulas is a feasible and precise method for targeted intracranial drug delivery.
- This robotic technique may improve surgical efficiency and accuracy for clinicians performing complex procedures involving multiple targeted brain infusions.
Overcoming the Blood-Brain Barrier via Convection-Enhanced Delivery
The blood-brain barrier remains the most significant physiological obstacle in neuro-oncology, preventing the vast majority of systemic chemotherapeutics and biopharmaceuticals from reaching therapeutic concentrations within malignant tissue [1, 2]. Convection-enhanced delivery addresses this challenge by utilizing a pressure gradient to drive therapeutics directly into the brain parenchyma, bypassing systemic limitations and reducing off-target toxicity [3, 4]. While early-phase trials for diffuse intrinsic pontine glioma and recurrent glioblastoma have demonstrated the safety of this approach, achieving an adequate volume of distribution often requires multiple infusion sites to cover infiltrative tumor margins [5, 6]. However, the manual stereotactic placement of several catheters can be time-consuming and technically demanding, potentially limiting the clinical scalability of multi-port infusion protocols [7, 8]. To overcome these logistical hurdles, researchers recently evaluated whether integrating robotic assistance with intraoperative CT guidance could streamline this complex delivery process.
Robotic Assistance in Multi-Target Surgical Simulation
To evaluate the feasibility, efficiency, and accuracy of robot-assisted, CT-guided infusion cannula placement, researchers utilized a high-fidelity simulation model. The study employed five 3D-printed phantom heads (anatomically accurate physical models used to simulate human surgical procedures) designed to replicate the structural challenges of neurosurgery. This experimental setup allowed the investigators to perform simultaneous bilateral placement of multiple cannulas, a technique intended to address the clinical need for broad therapeutic distribution in the treatment of infiltrative brain tumors. By using these phantoms, the team assessed the technical performance of the robotic system in a controlled environment that mimics the spatial constraints of the operating room. The study included a total of 30 targets, with six targets distributed within each of the five phantom heads. To ensure the findings translate to various real-world surgical scenarios, the researchers approached these targets from three distinct entry points, utilizing frontal, parietal, and occipital trajectories. Each of these three anatomical corridors was used for 10 targets, providing a comprehensive assessment of the system's versatility. This multi-target approach is highly relevant for neurosurgeons who must deliver therapeutics to diverse regions of the brain, demonstrating that the robotic system can maintain its performance regardless of the entry site or the complexity of the bilateral trajectory.
Stereotactic Accuracy Across Three Dimensions
The researchers confirmed the technical success of the procedure by verifying that all cannulas were clearly identified on intraoperative CT and MRI, ensuring that the robotic system successfully reached the intended anatomical regions. These targets were located at significant distances from the skull surface, with a mean target depth of 72.9 mm (SD 15.1, range 50.2 to 97.8 mm). This depth is clinically relevant because it reflects the real-world challenge of reaching deep-seated structures while maintaining a perfectly straight trajectory, an absolute requirement for effective convection-enhanced delivery. Accuracy was rigorously quantified across three planes using both imaging modalities. Based on CT imaging, the mean coronal error was 0.7 mm (SD 0.5, range 0.1 to 1.8 mm), the mean sagittal error was 1.0 mm (SD 0.7, range 0.0 to 2.7 mm), and the mean axial error was 0.6 mm (SD 0.5, range 0.1 to 1.8 mm). These measurements demonstrate sub-millimeter or near-millimeter precision in every dimension when verified against the intraoperative scan. To further validate these findings, the investigators performed post-placement MRI, which yielded comparable results. The MRI analysis showed a mean coronal error of 0.8 mm (SD 0.5, range 0.1 to 1.9 mm), a mean sagittal error of 1.3 mm (SD 1.1, range 0.0 to 5.2 mm), and a mean axial error of 0.9 mm (SD 0.7, range 0.1 to 2.7 mm). The study also calculated the radial error, a metric representing the shortest three-dimensional distance between the planned target point and the actual center of the cannula tip. The mean radial error on CT was 0.4 mm (SD 0.7, range 0.01 to 2.3 mm), while the mean radial error on MRI was 0.6 mm (SD 0.7, range 0.02 to 2.9 mm). For the practicing neurosurgeon, these low radial error values indicate that the robotic system can consistently place cannulas within a fraction of a millimeter of the intended target, minimizing damage to adjacent healthy brain tissue.
Clinical Efficiency and Procedural Implications
Beyond the high degree of stereotactic accuracy, the study demonstrated significant temporal efficiency, a critical factor in reducing anesthesia time and improving operating room throughput. The researchers reported that the time from initial target acquisition to the placement of all cannulas was less than 30 minutes per phantom. This rapid workflow is highly relevant for clinicians treating diffuse or multifocal pathologies, such as glioblastoma or other infiltrative tumors, where delivering therapeutic agents often requires the simultaneous placement of multiple cannulas to ensure adequate volumetric coverage of the tumor and its margins. By streamlining the trajectory planning and insertion process, this robotic method addresses the logistical challenges of multi-cannula convection-enhanced delivery, which has historically been limited by the time-intensive nature of manual stereotactic adjustments. The investigators concluded that the simultaneous CT-guided robotic insertion of multiple infusion cannulas was feasible, efficient, and precise. By integrating robotic assistance with real-time imaging, the procedure minimizes the manual adjustments and iterative imaging steps often required in traditional frame-based or frameless stereotaxy. For the practicing neurosurgeon, these findings suggest that the technique could improve surgical efficiency for complex procedures involving targeted drug delivery, potentially allowing for more standardized and reproducible outcomes. The ability to achieve sub-millimeter precision within a compressed surgical timeframe addresses a major technical barrier to the widespread clinical adoption of direct-to-parenchyma infusion therapies.
References
1. Wu D, Chen Q, Chen X, Han F, Chen Z, Wang Y. The blood–brain barrier: Structure, regulation and drug delivery. Signal Transduction and Targeted Therapy. 2023. doi:10.1038/s41392-023-01481-w
2. Wen PY, Weller M, Lee EQ, et al. Glioblastoma in adults: a Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) consensus review on current management and future directions. Neuro-Oncology. 2020. doi:10.1093/neuonc/noaa106
3. Fredricks N, Harkins M, Price AM, Morden F, Dewan MC. Protocols and safety profile of convection enhanced delivery in pediatric patients: a systematic review. Child s Nervous System. 2026. doi:10.1007/s00381-025-07121-8
4. Halle B, Mongelard K, Poulsen FR. Convection-enhanced Drug Delivery for Glioblastoma: A Systematic Review Focused on Methodological Differences in the Use of the Convection-enhanced Delivery Method.. Asian journal of neurosurgery. 2019. doi:10.4103/ajns.AJNS_302_17
5. Zacharoulis S, Dadario NB, DeWalt N, et al. Chronic convection-enhanced delivery of MTX110 into the brainstem for pediatric diffuse midline glioma, phase I trial. 2026. doi:10.1093/neuped/wuag001
6. Heiss JD, Jamshidi A, Shah S, et al. Phase I trial of convection-enhanced delivery of IL13-Pseudomonas toxin in children with diffuse intrinsic pontine glioma.. Journal of neurosurgery. Pediatrics. 2019. doi:10.3171/2018.9.PEDS17225
7. Vogelbaum MA, Brewer C, Barnett GH, et al. First-in-human evaluation of the Cleveland Multiport Catheter for convection-enhanced delivery of topotecan in recurrent high-grade glioma: results of pilot trial 1.. Journal of neurosurgery. 2019. doi:10.3171/2017.10.JNS171845
8. McCrea HJ, Ivanidze J, O'Connor A, et al. Intraarterial delivery of bevacizumab and cetuximab utilizing blood-brain barrier disruption in children with high-grade glioma and diffuse intrinsic pontine glioma: results of a phase I trial.. Journal of neurosurgery. Pediatrics. 2021. doi:10.3171/2021.3.PEDS20738